News
OSL Group’s Christmas lunch on December 18, 2025. It's nice to come together!

Our first book chapter is now online! It appears in Science of Synthesis: Modern Strategies in Organofluorine Chemistry 2, as Chapter 2: Synthesis of Monofluoromethylated Compounds.
Modern Strategies in Organofluorine Chemistry 2
2.2 Synthesis of Monofluoromethylated Compounds
The incorporation of monofluoromethyl groups into organic scaffolds has emerged as a prominent area of interest since the mid-20th century, driven by the unique biological properties these groups impart. This substituent functions as a versatile bioisostere for methyl and hydroxymethyl groups, with the capacity to form subtle hydrogen bonds that significantly impact molecular recognition and pharmacological behavior. In contrast to the well-established di- and trifluoromethylation reactions, monofluoromethylation remains synthetically challenging due to the inherent instability and high reactivity of monofluoromethyl reagents. Since the early 1980s, substantial efforts have been dedicated to developing reliable, cost-effective, and practical methodologies for monofluoromethyl group introduction. This review summarizes recent advances in reagent design and synthetic strategies for achieving selective and efficient monofluoromethylation, underscoring its growing importance in drug discovery and functional materials.
Read more: https://www.thieme-connect.de/products/ebooks/abstract/10.1055/sos-SD-244-00320
We are excited to welcome Rudolfs to our group!
New article in Advanced Synthesis & Catalysis 'Monofluorinated C1 Synthon Strategy for the Construction of Fluoromethylene-Linked Bicyclo[1.1.1]Pentane Derivatives' by Arturs & Toms. Great work!
Monofluorinated C1 Synthon Strategy for the Construction of Fluoromethylene-Linked Bicyclo[1.1.1]Pentane Derivatives
Fluoroalkyl-substituted bicyclo[1.1.1]pentanes (BCPs) have emerged as an attractive scaffold in drug discovery. Herein, the modular construction of fluoromethyl-linked BCPs is reported. Fluoroiodomethyl phenyl sulfoxide is found to be a synthetic equivalent of a formal fluoromethylene radical cation synthon, which, under metal-free conditions and violet light irradiation (400 nm), enables an atom-transfer radical addition reaction to [1.1.1]propellane. This straightforward approach provides access to novel bicyclo[1.1.1]pentane-substituted fluoromethyl sulfonium reagents. The electrophilic properties of these sulfonium salts allow nucleophilic displacement under mild conditions, enabling the introduction of the fluoromethyl bicyclopentyl group into diverse natural products and drug molecules with good functional group tolerance.
Read more: http://https://doi.org/10.1002/adsc.70119
The authors are grateful for the financial support from Recovery and Resilience Facility (5.2.1.1.i.) (grant no. 14/OSI/ZG).
OSL Group’s 3rd Anniversary at the Valmiermuiza Brewery. Good memmories!


We participated in the Researchers's Night 2025 as well on September 25–26, 2025. Crazy night!
This year, Janis, Renate, Estere, Andrei, Ram, Toms, Arturs, Violeta, Valerija and Jevgenijs was actively involved in educational outreach for children and the general public, conducting interactive experiments and leading laboratory tours.












More information: https://www.osi.lv/21237-2/zinatnieku-nakts-2024/
https://www.researchlatvia.gov.lv/lv/night-events/latvijas-organiskas-sintezes-instituts
Our group participated in the 14th Paul Walden Symposium, held on September 25–26, 2025, at the Tallink Hotel Riga.
Additionally, each year the Paul Walden Symposium provides an opportunity for two PhD students to present their doctoral research. This year, Arturs was selected to give an oral presentation on his PhD work "Fluorinated synthon transfer from sulfonium salts".

This year, four students from our group participated in the poster session:
Toms Pfeifers (Access to 2,2-fluoroiodobicyclo[1.1.1]pentanes)
Estere Zazerska (Synthetic access to fluorohalomethyl esters)
Maris Konisevs (Progress toward 1-fluoropropellane: synthesis of advanced [1.1.1]propellane intermediates)
.jpeg)
.jpeg)
.jpeg)
The poster prize for one of the best bachelor level poster awarded by the Latvian Institute of Organic Synthesis Fund, was received by B.Sc. student Toms Pfeifers (University of Latvia, LIOS): https://walden.osi.lv/14th-paul-walden-symposium/14th-paul-walden-symposium-awards/
More information about 14th Walden symposim program and speakers: https://walden.osi.lv/14th-paul-walden-symposium/
Take a look at the abstract book: https://walden.osi.lv/wp-content/uploads/2025/09/Walden-2025-Abstract-Book.pdf
Great news!! Anete is leaving our group to pursue doctoral studies at ETH Zürich, where she will join the research group of Prof. Dr. Helma Wennemers. We wish you a PhD journey full of success and many publications!

Check out her latest publications: https://www.scopus.com/authid/detail.uri?authorId=59913699100&origin=resultslist
Follow her on LinkedIn: https://www.linkedin.com/in/anete-patricija-raiskuma-26b5a1203/
New article in Advanced Synthesis & Catalysis 'Acridinium Photocatalyzed Monofluoromethyl Radical Cascade Reaction of Alkenes with Iodine(III) Reagent: A Mechanistic Study' by Anete & Ram. Impressive team work!!
Acridinium Photocatalyzed Monofluoromethyl Radical Cascade Reaction of Alkenes with Iodine(III) Reagent: A Mechanistic Study
Acridinium salts are well-known and highly effective organic photoredox catalysts, particularly recognized for their strong oxidizing properties. While they are widely used as photo-oxidants, their potential as photoreductants, formed easily through interactions with electron donors, has been largely overlooked and underexplored in photocatalysis. In this study, the application of an acridinium salt as a photocatalyst for the reductive activation of iodine(III) reagent containing monofluoroacetoxy ligands is described. This process allows for the synthesis of oxindoles and related N-heterocycles with monofluoromethyl groups, starting from alkenyl N-arylamides. Detailed mechanistic studies are conducted to gain insight into the in situ formation of the active acridine radical species, catalyst species interconversion, and the photocatalytic mechanism underlying the monofluoromethyl radical cascade reaction of alkenyl N-arylamides. A wide range of mechanistic tools is employed, including radical trapping experiments, UV–Vis absorption spectroscopy, time-resolved photoluminescence quenching experiments, electron paramagnetic resonance spectroscopy, and density functional theory calculations. The synthetic utility of this protocol is demonstrated through a substrate scope study, which highlights the efficient access to oxindoles and other polycyclic heterocycles featuring monofluoromethyl units which are widely recognized for their biological significance.
Read more: https://doi.org/10.1002/adsc.70069
This work has been supported by Recovery and Resilience Facility (5.2.1.1.i.) (grant no. 14/OSI/ZG), Latvian Institute of Organic Synthesis Internal (grant no. IG-2023-11), and Estonian Research Council (grant no. PSG828).
New article in Organic Letters 'Metal-Catalyzed Fluoroacetyl Carbene Transfer from Sulfonium Salts' by Arturs & Janis. Nice work!
Metal-Catalyzed Fluoroacetyl Carbene Transfer from Sulfonium Salts
The synthesis of organofluorine compounds is pivotal in developing innovative pharmaceuticals, materials, and agrochemicals. Fluorinated carbene transfer offers a promising strategy for forming new carbon–carbon and carbon–heteroatom bonds, facilitating one-carbon editing by simultaneously introducing fluorine into target structures. In this study, we introduce a novel fluoroacetyl sulfonium reagent, (2-ethoxy-1-fluoro-2-oxoethyl)(phenyl)(2,3,4,5-tetramethylphenyl)sulfonium tetrafluoroborate, serving as an effective alternative to the currently unknown 2-diazo-2-fluoroacetate for ethyl fluoroacetyl carbene transfer. This reagent is applied in Doyle–Kirmse and cyclopropanation reactions operating under mild conditions with the use of earth-abundant metal catalysts. This approach enables the efficient synthesis of valuable monofluorinated products.
Read more: https://doi.org/10.1021/acs.orglett.5c01416
This work has been supported by Latvian Council of Science Project lzp-2022/1-0335.
Great News! Amazing article in JACS 'Monofluoromethyl Radical Mediated Halogen-Atom Transfer' by Ram & Anete. Outstanding!
Monofluoromethyl Radical Mediated Halogen-Atom Transfer
Halogen-atom transfer (XAT) is an effective method for generating carbon radicals from alkyl halides. In this work, we present a novel XAT method that eliminates the need for photocatalysts by utilizing the direct excitation of a monofluoroacetoxy ligand-containing hypervalent iodine(III) reagent. This approach allows nucleophilic monofluoromethyl radicals to efficiently form carbon radicals from activated alkyl halides. These radicals can subsequently functionalize unactivated alkenes through atom transfer radical addition (ATRA). Our mechanistic studies provide insights into the generation of monofluoromethyl radical and its unique reactivity. This work highlights the untapped potential of simple, single carbon atom-containing radicals for effective alkyl radical generation.
Read more: https://pubs.acs.org/doi/10.1021/jacs.5c08589
This work has been supported by RRF grant No. 38/OSI/PG (RRF project No. 5.2.1.1.i.0/2/24/I/CFLA/001).
This summer we are excited to welcome to our group two new PostDocs - Dr. Ankit Kumar & Dr. Anupam Das!
Dr. Ankit Kumar has started working on the HORIZON-WIDERA-2023-TALENTS-02-01 project titled 'De novo Synthesis of Fluoroarenes and Fluoro(hetero)arenes (FARAON)' (Project ID: 101180677).
Dr. Anupam Das has joined the Refinanced ERC Starting Grant project via the European Regional Development Fund, titled 'Fluorinated Compounds by Nodal Synthesis (F-NODE)' (Grant No. 1.1.1.5/2/24/A/001).
Our colleagues are presenting their exciting research at the 23rd European Symposium on Organic Chemistry (ESOC, Copenhagen, June 29th-July 3rd 2025)
During the poster session, Arturs and Toms disclosed the synthesis of novel bicyclo[1.1.1]pentane-containing sulfonium salts starting from fluoroiodomethyl sulfoxide. The application of these reagents was demonstrated for the mild transfer of fluoromethyl bicyclo[1.1.1]pentane to various nucleophiles under basic conditions.
Meanwhile, Ram demonstrated a novel XAT method based on the direct excitation of a monofluoroacetoxy ligand-containing hypervalent iodine(III) reagent, eliminating the need for photocatalysts. This approach efficiently generates nucleophilic monofluoromethyl radicals and enables the formation of carbon radicals from activated alkyl halides. These radicals can subsequently functionalize unactivated alkenes via atom transfer radical addition (ATRA).
Good vibes and great science at the poster session >>>
.jpeg)


More info about the programme and poster sessions: https://discongress.eventsair.com/2503-esoc-2025
Anete successfully defended her Master’s thesis, “Carbon-Centred Radical Mediated Halogen Atom Transfer Reaction”, at the University of Latvia. 🎓Congratulations! (03/06/25)
In her thesis, she explored how carbon-based radicals can be efficient halogen atom transfer (XAT) reagents. Monofluoromethyl radicals, due to their nucleophilic nature, can easily abstract halogen atoms from activated alkyl halides. This work presents a promising alternative to traditional XAT methods using toxic tin radicals or photocatalysis, thanks to a novel route using direct excitation of a monofluoroacetoxy ligand-containing hypervalent iodine(III) reagent.
To find out more check out the preprint of her work: https://chemrxiv.org/engage/chemrxiv/article-details/681d4b7d50018ac7c5130c56


It has been proven that ASAP publications are more effectively discussed over a glass of beer!


Our group's Spring hike was a great opportunity to discuss chemistry in nature!









Our group's researchers and students participated in the Shadow day 2025 on 4th April.









Shadow Day's participants:
Renate Vaisgerberga was shadowed by Gundars Agarskis (Jelgava Spidola State Gymnasium 10th grade)
Anete Patricija Raiskuma was shadowed by Dainis Bivka (Daugavpils Technology High School – Lyceum 10th grade)
Arturs Sperga was shadowed by Kristers Urtans (Riga Janis Sreinhauers High School 10th grade)
Janis Zakis was shadowed by Matvejs Ogurcovs (Riga State Classical Gymnasium 10th grade)
Ram Kumar Nagarajan was shadowed by Karlis Bumbahs (Talsi State Gymnasium 11th grade)
Andrei Baran was shadowed by Tomass Matusevics (Riga State 3rd Gymnasium 11th grade)
More info: https://www.osi.lv/enu-dienai-4-aprilim-esam-atverusi-30-vakances/
https://enudiena.lv/ed/par-mums
Congrats, Renate, on receiving the The Latvian Academy of Sciences (LAS) and the Institute of Organic Synthesis (LIOS) Development Fund's Edmunds Lukevics Award! Impressive!!
The Latvian Academy of Sciences and the Latvian Institute of Organic Synthesis Development Fund's Edmunds Lukevics Award for 2025 is granted to Ph.D. Renāte Vaisgerberga (Institute of Organic Synthesis, LOSI) for her work "Development of Fluoromethylene Group Transfer Reagents," which was developed under the supervision of LAS Corresponding Member Dr. sci. nat. J. Veliks.
Ph. D. Renate Vaisgerberga has been working at the Institute of Organic Synthesis in Latvia since 2015. She obtained her Bachelor's, Master's, and Doctoral degrees from Riga Technical University. In her involvement in scientific work under the leadership of Dr. sci. nat. Janis Veliks, Renate focused on research in organic synthesis methodology, specifically the possibilities of introducing fluoromethylene groups into organic compound molecules.
Although fluorine atoms are already found in the structure of every fifth active pharmaceutical ingredient, the range of synthetic methods for their introduction remains quite limited, often requiring the use of aggressive and polluting reagents. Therefore, the relevance of Ph. D. Vaisgerberga’s research and its impact on the development of organic synthesis methodology is clear. The content of Renate Vaisgerberga's doctoral dissertation is reflected in seven scientific publications, and the young scientist has presented papers at several scientific conferences.
Currently, Ph. D. Vaisgerberga continues her work at the Organic Synthesis Laboratory at the Institute of Organic Synthesis, where she independently leads the research project "Novel Source Towards Challenging Monofluorocarbene."

More about Young Researcher Award recipient: https://www.lza.lv/images/LM_LZA%20balvas/LZA_balvas_2025/Edmunds_Lukevics_2025.pdf
We are excited to welcome three new members - Valerija, Maris and Estere to our group!
Congrats, Laura, on receiving the Young Researcher Award from the Latvian Academy of Sciences! Your hard work and dedication are truly inspiring! Wishing you continued success in all your future research and endeavors!
During her bachelor's studies, Laura began working at the Organic Synthesis Methodology Group at the Latvian Institute of Organic Synthesis and continues her work in the Organic Synthesis Laboratory under the guidance of Dr. sci. nat. Janis Veliks. In 2022, she earned a Bachelor of Science degree in Chemistry at the University of Latvia with the thesis “Synthesis of New AcrAB-TolC Efflux Pump Inhibitors” (supervisors: Dr. sci. nat. Janis Veliks, Dr. Marina Madre). In 2024, she completed her Master’s degree in Natural Sciences with the thesis “Synthesis of N-Heterocycle-containing Amino Acids” (supervisors: Dr. sci. nat. Janis Veliks, Dr. Marina Madre). Her research focuses on the inhibition of the SARS-CoV-2 nsp10/nsp16 2′-O-methyltransferase complex as a potential approach to the development of new antiviral agents, synthesizing N-heterocycle-containing amino acids that can serve as inhibitors and provide data for artificial intelligence-driven drug development.

More about Young Researcher Award recipient: https://lza.lv/images/LM_LZA%20balvas/LZA_balvas_2025/JZ_2_2025.pdf
Congratulations, Anete, on receiving the prestigious Mundheim Family Ecxellence scholarship from the University of Latvia Foundation!
.jpeg)
In 2013, the Mundheim family established the Excellence Scholarship, awarded to the top student from the University of Latvia’s bachelor’s programs in Biology or Chemistry who chooses to continue their studies at the master’s level.
More info about Mundheim family scholarship recipients: http://fonds.lv/scholarships/25
More about Anete: http://fonds.lv/students/911
Pictures from: https://www.facebook.com/share/p/1H2NNRpX4t/?mibextid=wwXIfr
https://www.facebook.com/share/p/1WVrGcApbn/?mibextid=wwXIfr
Wishing you a Merry Christmas and a Happy New Year!

We are improving problem solving skills together! #Goup event 2024

See more information about quest here: https://escapetown.lv/zelta-armijas-eliksirs/
Exciting news! We have 2 new group members joining us!
This year, Janis Mikelis Zakis received his PhD from the University of Strasbourg (France) and his doctoral thesis was on the topic: Cyclometallated iridium catalysts for ortho-directed C–H borylation. More info: https://www.scopus.com/authid/detail.uri?authorId=57424752300
Last year, Toms developed a high school research project in the OSL laboratory under Arturs supervision; this year, he has returned to the group as a first-year chemistry student of the Faculty of Natural Sciences and Technology.
New article in Organic Letters about novel diaryl fluorohalomethyl sulfonium salts and their application by Arturs & Janis. Well done!
Fluorohalomethylsulfonium Salts as a Fluorohalocarbene Source
Herein, we report efficient synthesis of novel diaryl fluorohalomethyl sulfonium salts. The application showcased proficient reagents for Freon-free fluorochloro-, fluorobromo-, and fluoroiodocarbene transfer to alkenes. The developed mild, room temperature conditions for the cyclopropanation do not require any metal catalysts and allow synthetic access to diverse cyclopropanes.
Read more: https://doi.org/10.1021/acs.orglett.4c02327
Acknowledgement: Latvian Council of Science project lzp-2022/1-0335
Our group members attended the BOS 2024 Conference (July 7-10, Riga) and presented their research during a poster session.
Conference programme: https://boschem.eu/bos2024/schedule/
Conference abstract book: https://boschem.eu/bos2024/abstract-book/
Posters were presented by:
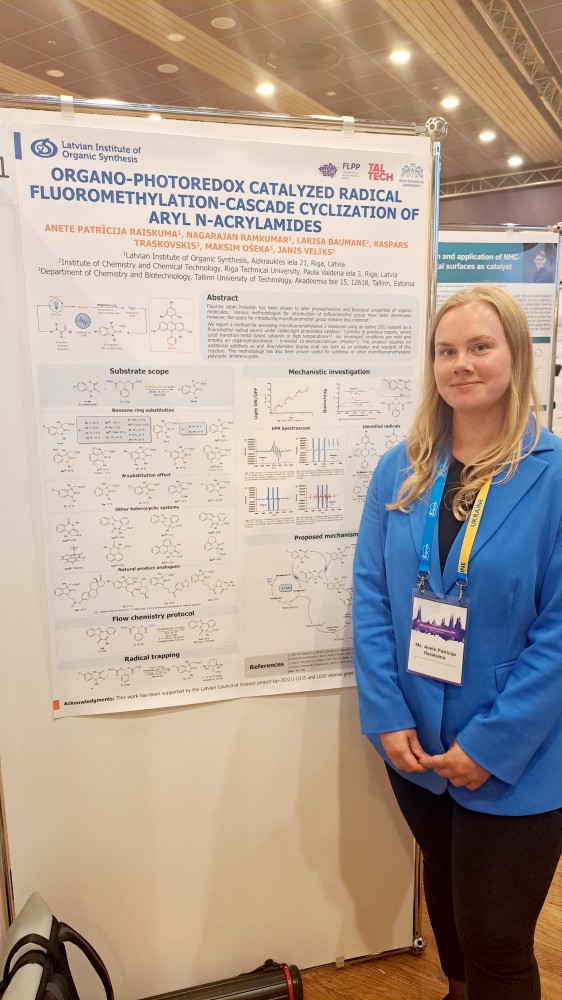
Nagarajan Ramkumar Arturs Sperga Laura Paunina Anete Patricija Raiskuma
and Ketrina Plantus
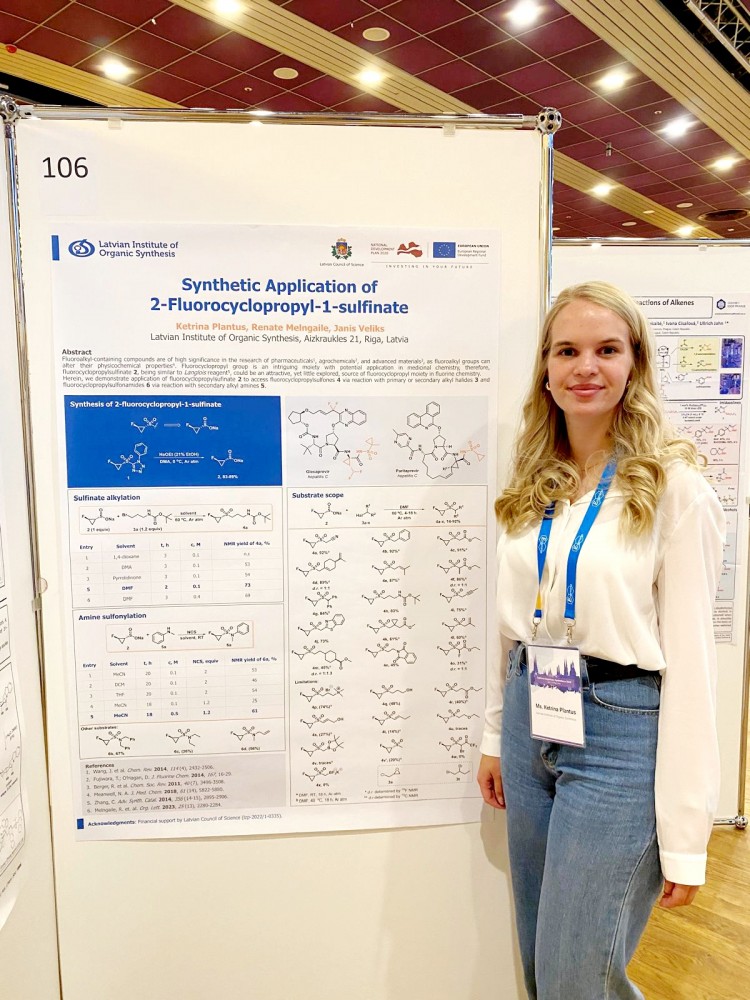
Ketrina has received her Bachelor's degree! Nicely done!
Ketrina has defended her Bachelor's Thesis "Synthetic application of 2-fluorocyclopropyl-1-sulfinate" at the Riga Technical University (12.06.2024.).
Congratulations on the first milestone in the career of a chemical scientist!
Laura has received her Master's degree! Congrats!
Laura has defended her Master's Thesis “Synthesis of amino acids containing N-Heterocycles” at the University of Latvia (07.06.2024.).
Congratulations and good luck in starting your PhD studies!
The award ceremony for winners of the Latvian Academy of Sciences competition - The Best Achievements in Latvian Science in 2023 was held on February 23, 2024.
The prize was given for research in applied science: ''Innovative reagent that significantly expands the possibilities for chemists to incorporate fluorine atoms''.
Authors: LAS corresponding member Janis Veliks, Ph.D. Nagarajan Ram Kumar, Dr.chem. Larisa Baumane, Dr.chem. Dzintars Zacs. #Latvian Institute of Organic Synthesis.
Video recording of the award ceremony: https://youtu.be/ksglFEJvWNE?si=2Chjfdz9KmsjlY_l
Wow! Janis received The Thieme Chemistry Journals Award ! #2024
Since 1999 – when the award was given for the first time to young researchers – it is its aim to send a sign of recognition and career encouragement to the new generation of organic chemists.
https://www.thieme.de/en/thieme-chemistry/thieme-chemistry-journals-award-107359.htm
We are happy to announce that Janis has been selected as the scientist of the month of September in the calendar #ZinātneLatvijai 2024!
Research as a creatively intuitive process. The calendar “Research Latvia 2024” – stories about 12 passionate personalities, outstanding in their field and endowed with unlimited creativity. Scientists help find solutions to global challenges and everyday problems through their research.
http://youtu.be/jKLoX-2vv2c?si=gFMd8dBRatCpjtkz
Renate has received The Emīlija Gudriniece Prize of Latvian Academy of Sciences – for the best work in chemistry or technology of chemistry in 2023!
Congratulations to Ph.D. Renate Melngaile for receiving The Emīlija Gudriniece Prize of Latvian Academy of Sciences on her Doctoral Thesis "Development of Fluoromethylene Transfer Reagents".
Dr.sc.nat Janis Veliks, Ph.D. Nagarajan Ram Kumar, Dr.chem. Larisa Baumane, Dr.chem. Dzintars Zacs - winners of the Latvian Academy of Sciences annual award “ACHIEVEMENTS IN SCIENCE 2023”
Researchers led by Dr.sc.nat. Janis Veliks have developed a new, stable, and non-aggressive reagent. In the presence of a catalyst and under the influence of blue LED light, this reagent generates fluoromethyl radicals – active particles containing fluorine that readily attach to organic molecules. This innovative reagent significantly expands the possibilities for chemists to incorporate fluorine atoms, paving the way for the construction of potential new drugs and the development of novel technologies for obtaining well-known substances.
Congratulations to Renate for receiving PhD degree!
Renate has successfully defended her PhD thesis! During doctoral studies she has been working on development of fluoromethylene transfer reagents and methodologies to access novel monofluorinated molecules. Congratulations and warmest regards from Veliks group!
Congratulations to Renate for her paper in OL!
Read our latest work in OL: Synthetic Access to Fluorocyclopropylidenes
Efficient reagent and methodology reported to access rare monofluorinated building-blocks - fluorocyclopropylidenes.
Arturs has received Young Researchers Award 2023 of Latvian Academy of Sciences!
Congratulations to Arturs for receiving Young Researchers Award 2023 of Latvian Academy of Sciences on his work on Iron Catalyzed Fluoromethylene Transfer.
http://www.lza.lv/aktualitates/jaunumi/1448-lza-jauno-zinatnieku-balvu-laureati-2023-arturs-sperga
Check out his ineterview on Latvian 1st Channel:
https://www.osi.lv/pasniegtas-lza-balvas-jaunajiem-zinatniekiem-tostarp-tris-osi-kimikiem/
Prime minister visits LIOS
Presenting infrastructure available at our group to Latvian prime minister Mr. K. Kariņš.
Congratulations to Ram for his paper in ACIE!
Read our latest work in ACIE: Merging Copper(I) Photoredox Catalysis and Iodine(III) Chemistry for the Oxy-monofluoromethylation of Alkenes
[Bis(monofluoroacetoxy)iodo]benzene- a new reagent for radical monofluoromethylation under visible-light copper(I) photoredox catalysis.
https://onlinelibrary.wiley.com/doi/10.1002/anie.202219027?af=R
Organic Synthesis Laboratory - New reasearch group at LIOS
We are glad to announce that Organic Synthesis Laboratory at LIOS starts working from 1st of October
Arturs won the best poster award of 12th Paul Walden Symposium
Arturs Sperga has participated in 12th Paul Walden Symposium on organic chemistry on 28-29th of October.
He won the award for the best poste for master degree students. Congratulations!